
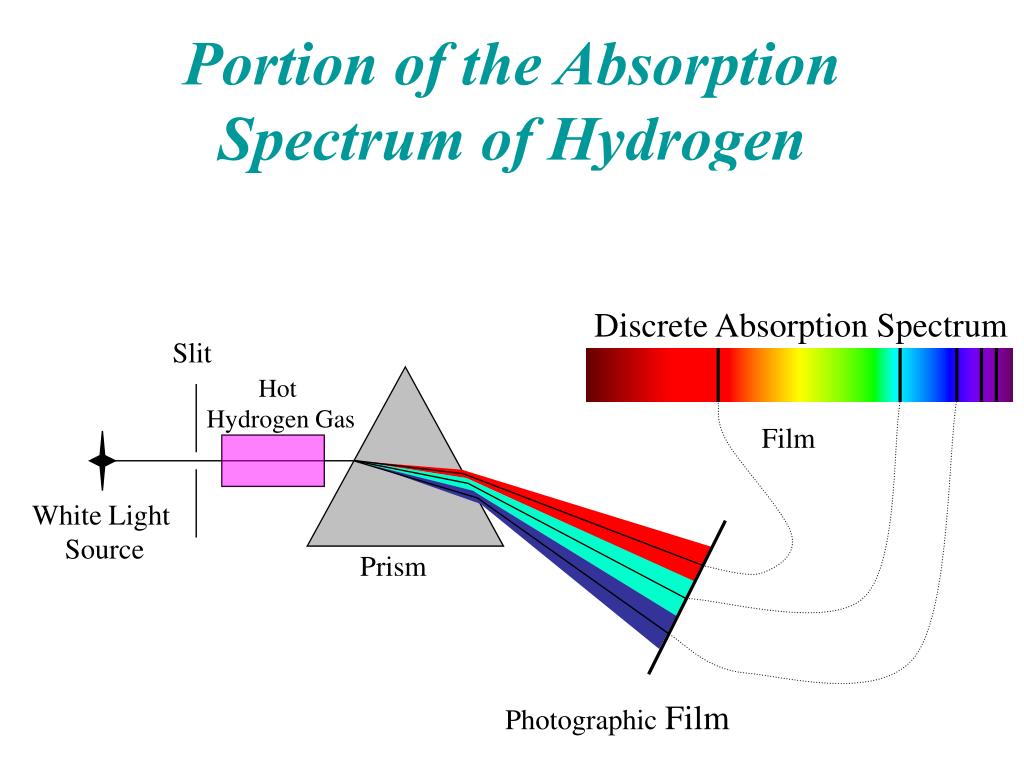
When the heating is turned off, the atoms revert to their ground state by emitting radiation of a specific wavelength. When an atom in its vapor state is heated to a high temperature, the electrons get excited. This behavior of atoms produces a series of dark lines on a background called atomic absorption spectra. The light of other wavelengths gets transmitted. When an atom in its vapor state is placed in white light, it absorbs light of characteristic wavelength due to the electrons present in it. There are two types of atomic spectra: Absorption spectra A dark area separates these spectral lines from one another. Due to their distinct orbitals or energy levels, all atoms produce line spectra or discontinuous spectra. The spectra, in fact, are energy deals of electrons.
0 Comments
Leave a Reply. |


 RSS Feed
RSS Feed
